
New available paresthesia-free waveforms, such as burst, high frequency or high dose, appear to limit this problem by suppressing patient perception of the therapy. , lead positioning can dramatically impact paresthesia coverage and energy consumption. This lack of selectivity, associated with the type of implanted lead, its positioning, its properties, patient variability and the challenge to provide adequate spatial and temporal neural targeting using electrical source-specific capabilities, can lead, on the one hand, to feel paresthesia in a non-painful area and, on the other hand, to not be able to cover decisive painful body regions and/or pain trigger(s), particularly in focal distal pain distribution, as in foot and groin pain. Despite its effectiveness, SCS has some limitations.įirst, pauci-discriminative stimulation of billions of sensory epicritic fibers of the dorsal columns using electrical impulses makes it very complex for SCS to be selective in terms of pain coverage for patients. Using this reversible treatment option, the prerequisite to address pain with conventional therapy modality is to obtain appropriate coverage of the painful area, by inducing paresthesia on corresponding painful territories.
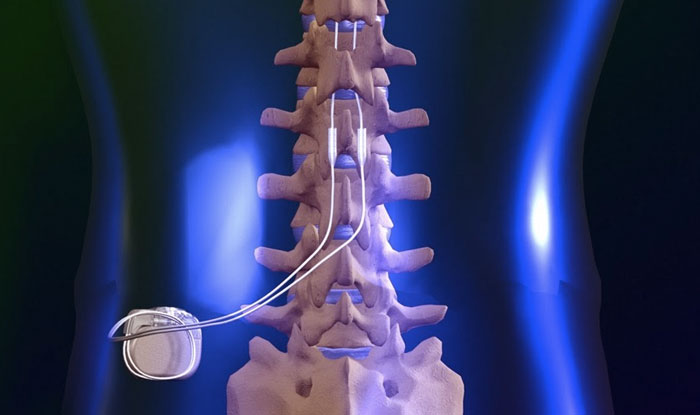
Spinal cord stimulation (SCS) is a well-established therapy to alleviate severe intractable neuropathic pain by improving pain control and quality of life in patients with failed back surgery syndrome (FBSS) or persistent spinal pain syndrome type 2 (PSPS-T2). In addition to our primary outcome based on pain rating, this study is designed to assess quality of life, functional disability, psychological distress, pain surface coverage, global impression of change, medication quantification, adverse events, brain functional imaging and electroencephalography, with the objective being to provide a multidimensional insight based on composite pain assessment. After 3 months, stimulation will consist in switching to burst for 1 month, and patients will choose which modality/waveform they receive and will then be reassessed at 6 and 12 months. Before crossing over, patients will receive each stimulation modality for 1 month, using tonic conventional stimulation. Sixty-six PSPS-T2 patients will be recruited internationally in three centers. “BOOST DRG” is the first prospective, randomized, double-blinded, crossover study to compare SCS vs. Comparisons between DRGS and standard SCS, or their combination, have never been studied on the same patients. As alternatives, new waveforms, such as burst stimulation and different spatial neural targets, such as dorsal root ganglion stimulation (DRGS), have shown promising results. While spinal cord stimulation (SCS) is a well-established therapy to address refractory persistent spinal pain syndrome after spinal surgery (PSPS-T2), its lack of spatial selectivity and reported discomfort due to positional effects can be considered as significant limitations.
